SUT Assemblies & Custom Tube Kits
SUT Assemblies & Custom Tube Kits for the Biopharmaceutical Industry
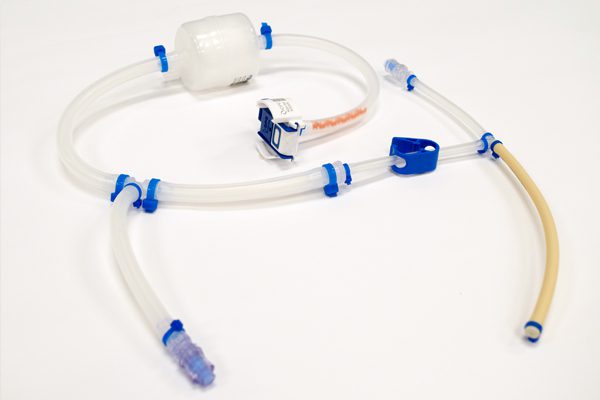
Reduce cross-contamination risks. Simplify cleaning procedures. Customize solutions. Single-Use Technology (SUT) assemblies address all of these issues and have emerged as a key manufacturing platform for the global biopharmaceutical community.
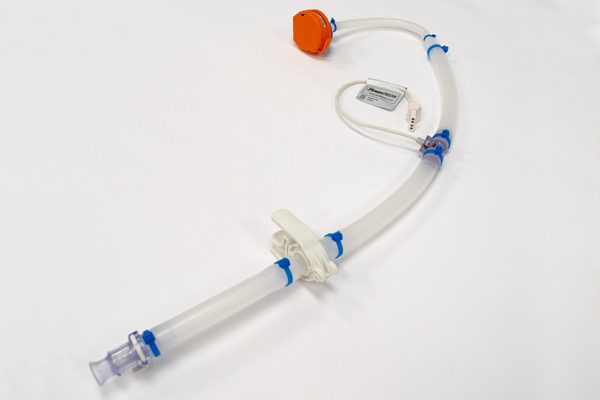
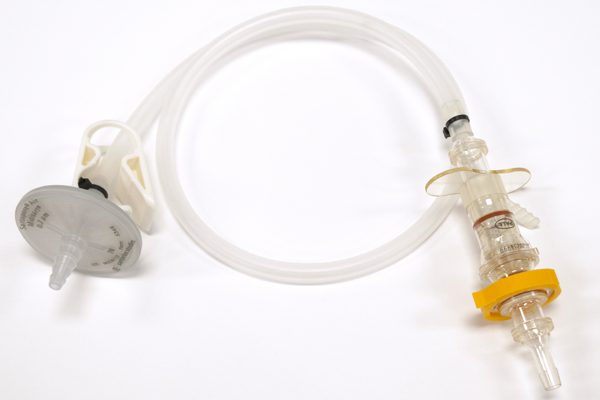
WHK BioSystems’ single-use consumables, including single-use manifolds, aseptic transfer bottle assemblies, and custom tube kits, streamline upstream processes while protecting the quality you’re accustomed to. Regardless of your company’s unique process, a variety of products and components can be assembled to meet your specific needs.
With a large inventory of commonly used components in stock, we are able to offer competitive lead times with quick delivery of goods. Plus, we are continually developing our supply chain flexibility to provide you with single-use solutions as efficiently as possible.
Providing Solutions Tailored to Cell & Gene Therapies
Short Lead Times
With a large component inventory available, we can quote in days, not weeks or months.
Extensive Library
A tooling library in Silicone and TPE means we can produce what you want, when you need it.
Experienced Teams
Our teams have the material and process expertise to handle the most demanding requirements.
Customized Solutions for Every Process
Our bottle assemblies accommodate most standard bottle sizes and offer a variety of configurations.
|
|
|
A well-designed tube set helps you increase productivity while reducing operating costs and contamination risks. It also meets the demands of the life science, biotech, and medical industries.
|
|
  |
Cell and gene therapy processes can vary widely, and custom, single-use manifolds can help increase productivity, reduce cost, and aid with scalability. Choose for a variety of arrangements, including:
|
|
  |
A core competency of WHK, injection molded and overmolded solutions provide the cleanest, most secure fluid management products in the industry.
|
|
  |
Downloadable Infosheets
Single-Use Technology by Design
Unique Processes Call For Custom Kits
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
FEATURES
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
APPLICATIONS
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
![]()
BENEFITS
Lessen Cross-Contamination Risks
Components can arrive sterile and go straight to disposal after a single use.
Simplified Cleaning Procedures
Single-use components do not require cleaning and sanitizing like Stainless-Steel systems.
Improved Efficiencies
Single-use technology allows you to proceed from batch to batch without excessive downtime due to sanitizing.
Experienced Team
Engineering team has material and processing expertise to handle demanding requirements. Design Team with hi-tech labs allows us to design products without losing sight of cost, quality, or delivery.
Quality Manufacturing
ISO 13485 quality management system, Class 7 cleanroom manufacturing, Decades of regulatory and industry knowledge.
Cell and Gene Therapies
The emergence of cell and gene therapies has created unique fluid-handling challenges for traditional SUT suppliers. WHK specializes in manufacturing complex tube kits that address these particular applications.
-
Development Support
-
Short Lead Times
-
ISO 13485 Certified
-
Class 7 Cleanrooms
-
Injection Molding
-
Low Order Minimums
-
ISO 11137 Sterility
-
Integrity Testing Services
LET'S TALK
Tell us your challenges, and together we will find a solution.


Disclaimer: User is responsible for determining suitability and safety of all products for intended use. Information as supplied on this site is intended to provide guidance only. WHK BioSystems disclaims all liability regarding product fitness for use. WHK BioSystems has also relied on raw material suppliers for a portion of the information and compliance statements contained on this site.
WHK BioSystems, LLC, maintains an ISO 13485:2016 registered quality management system. It operates Class 7 cleanroom manufacturing facilities.