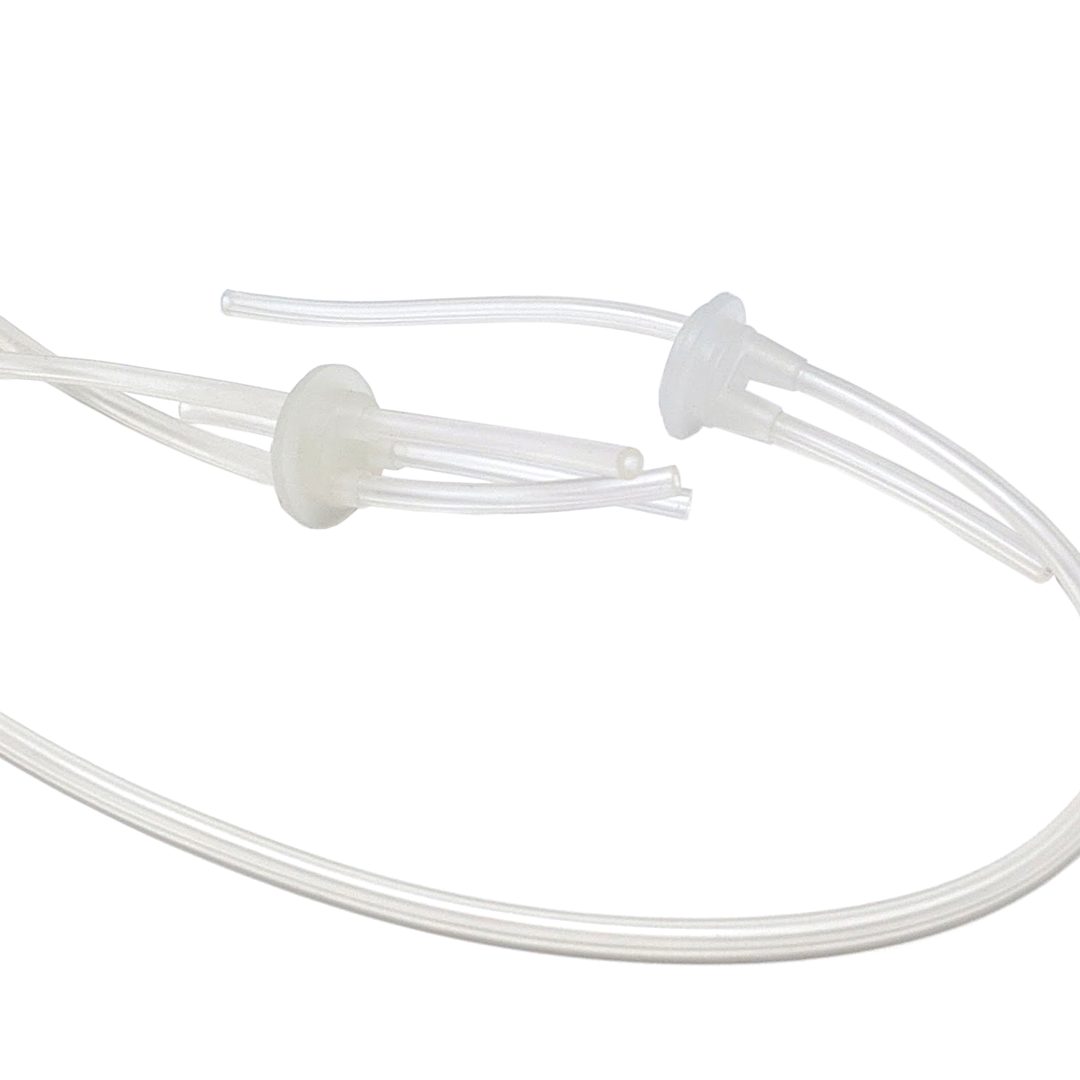
Injection Molding & Overmolding
Cleanroom Injection Molding & Overmolding
WHK BioSystems is a global manufacturer of high-quality injection molded and overmolded components for the biopharmaceutical and life science industries. From product design and process validation to quality parts and custom packaging, we can fulfill your project requirements
Why Choose WHK?
There are thousands of “Medical Molders” out there to choose from, but many of these same companies are primarily focused on industrial products and the more relaxed quality and regulatory standards those applications require.
WHK Biosystems specializes in medical-grade applications and our regulatory experience, quality, and attention to detail set us apart from the rest.
Capable. Compliant. Quality.
With two, well-equipped Class 7 cleanroom manufacturing facilities, we have space and capacity to take your project from development to large-scale production. Whether your job is a small batch prototype or involves an ongoing mass production contract, we can meet your needs.
Injection-molded and overmolded medical device components are often used in combination with our W-TPE™ Tubing to create many types of Single-Use Systems for the biopharmaceutical and life sciences industries. All components are compliant with the standards set by the healthcare, medical, and biopharmaceutical industries.
CAPABILITIES
- Process validation from project initiation through to completion
- Injection molding and overmolding of parts performed in fully-enclosed, Class 7 cleanrooms
- Quality assurance maintained by ISO 13485:2016 certification
- Product design and custom packaging tailored to your needs
- Real-time process monitoring
- Full library of medical-grade materials
MATERIALS
- Polypropylene (PP)
- Liquid Silicone Rubber (LSR)
- Thermoplastic Elastomers (TPE)
- Polycarbonate (PC)
- Polyethylene (PE)
- Polyether Sulfone (PES)
- Thermoplastic Polyurethane (TPU)
- Polyvinylidene Fluoride (PVDF)
- Acrylonitrile Butadiene Styrene (ABS)
Let’s Chat
Your unique processes call for customizable solutions.
Tell us your challenges and together we will come up with a solution.

Disclaimer: User is responsible for determining suitability and safety of all products for intended use. Information as supplied on this site is intended to provide guidance only. WHK BioSystems disclaims all liability regarding product fitness for use. WHK BioSystems has also relied on raw material suppliers for a portion of the information and compliance statements contained on this site.
WHK BioSystems, LLC, maintains an ISO 13485:2016 registered quality management system. It operates Class 7 cleanroom manufacturing facilities.